Don't have an account?
Adverse events should be reported.
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.
Adverse events should also be reported to GE HealthCare at
gpv.drugsafety@gehealthcare.com
Adverse events should be reported.
Reporting forms and information can be found at https://yellowcard.mhra.gov.uk/.
Adverse events should also be reported to GE HealthCare at
gpv.drugsafety@gehealthcare.com
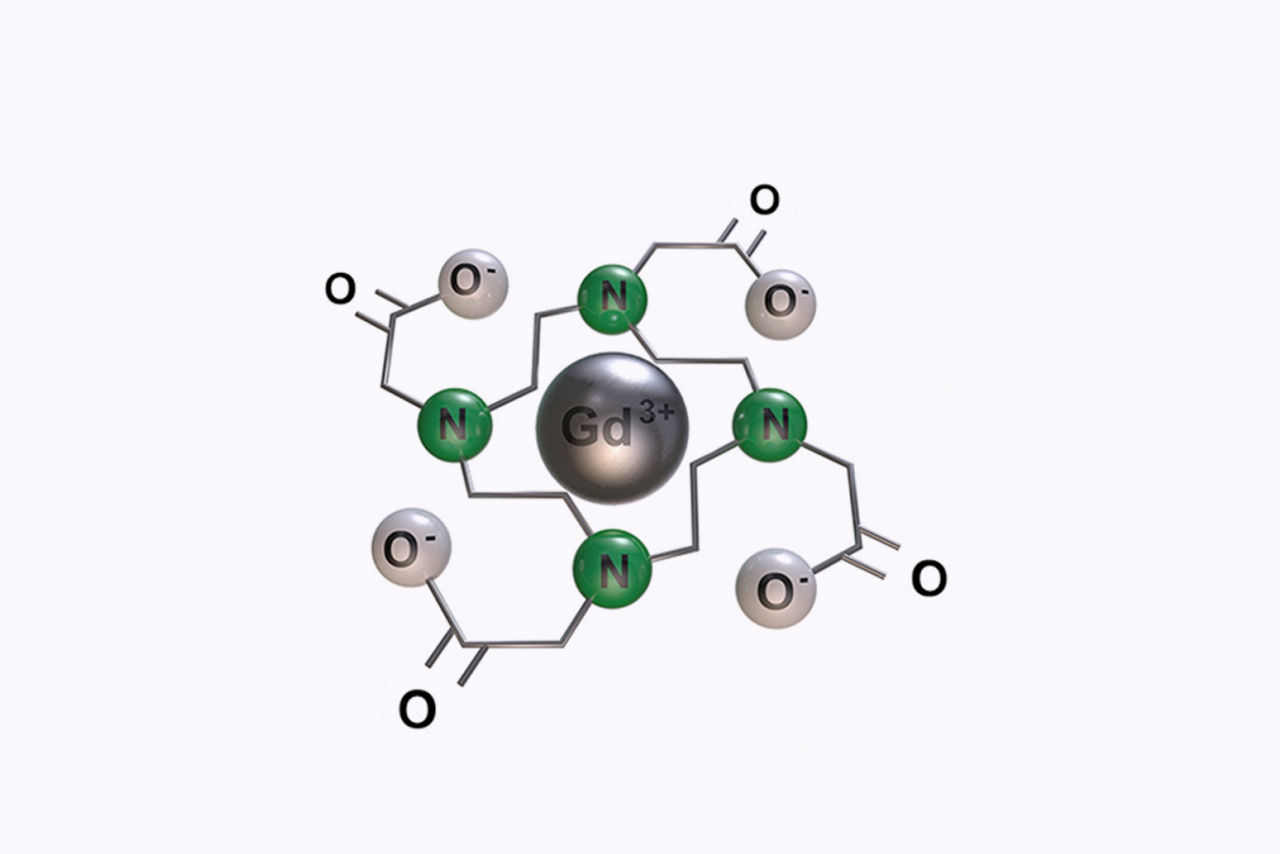
Macrocyclic structure
Macrocyclic, ionic chelate provides high kinetic and thermodynamic stability1
Expertly manufactured
Quality manufacture and delivery, expertly managed by GE HealthCare for greatest control
Established tolerability
Well-tolerated, with over 34 million patient administrations2,3
Convenient packaging options
Vials, prefilled syringes and +PLUS PAK bottles in a range of volumes
Clariscan is indicated for diagnostic contrast enhancement in3:
Clariscan should be used when diagnostic information is essential and not available with unenhanced magnetic resonance imaging (MRI).
Gadoteric acid must not be used intrathecally. Serious, life-threatening and fatal cases, primarily with neurological reactions (e.g. coma, encephalopathy, seizures), have been reported with intrathecal use. For intravenous injection only.
Clariscan offers a range of packaging:
Our unique +
PLUS
PAK packaging reduces hazard and downtime:
It also reduces cost and environmental impact:

There are no signals to suggest that the safety profile of Clariscan is different to that established for the reference product, Dotarem®.2
Hypersensitivity reactions: As with other intravenous contrast agents, gadoteric acid can be associated with anaphylactoid / hypersensitivity or other idiosyncratic reactions, characterised by cardiovascular, respiratory or cutaneous manifestations, and ranging to severe reactions including shock. In general, patients with cardiovascular disease are more susceptible to serious or even fatal outcomes of severe hypersensitivity reactions. The risk of hypersensitivity reactions may be higher in case of:
In patients with an allergic disposition the decision to use Clariscan must be made after particularly careful evaluation of the risk-benefit ratio. Most of these reactions occur within half an hour of administration. Therefore, post-procedure observation of the patient is recommended. Medication for the treatment of hypersensitivity reactions as well as preparedness for institution of emergency measures are necessary. Delayed reactions (after hours up to several days) have been rarely observed.
Impaired renal function: Prior to administration of Clariscan, it is recommended that all patients are screened for renal dysfunction by obtaining laboratory tests.
There have been reports of nephrogenic systemic fibrosis (NSF) associated with use of some gadolinium-containing contrast agents in patients with acute or chronic severe renal impairment (GFR< 30 mL/min/1.73m2). Patients undergoing liver transplantation are at particularly high risk since the incidence of acute renal failure is high in this group
Elderly: As the renal clearance of gadoteric acid may be impaired in the elderly, it is particularly important to screen patients aged 65 years and older for renal dysfunction.
Neonates and infants: Due to immature renal function in neonates up to 4 weeks of age and infants up to 1 year of age, Clariscan should only be used in these patients after careful consideration.
Cardiovascular disease: In patients with severe cardiovascular disease Clariscan should only be administered after careful benefit assessment because only limited data are available so far.
For full safety information, including information on use in pregnancy and breast-feeding women, please consult the Clariscan SmPC.

Dotarem is a trademark of Guerbet LLC.