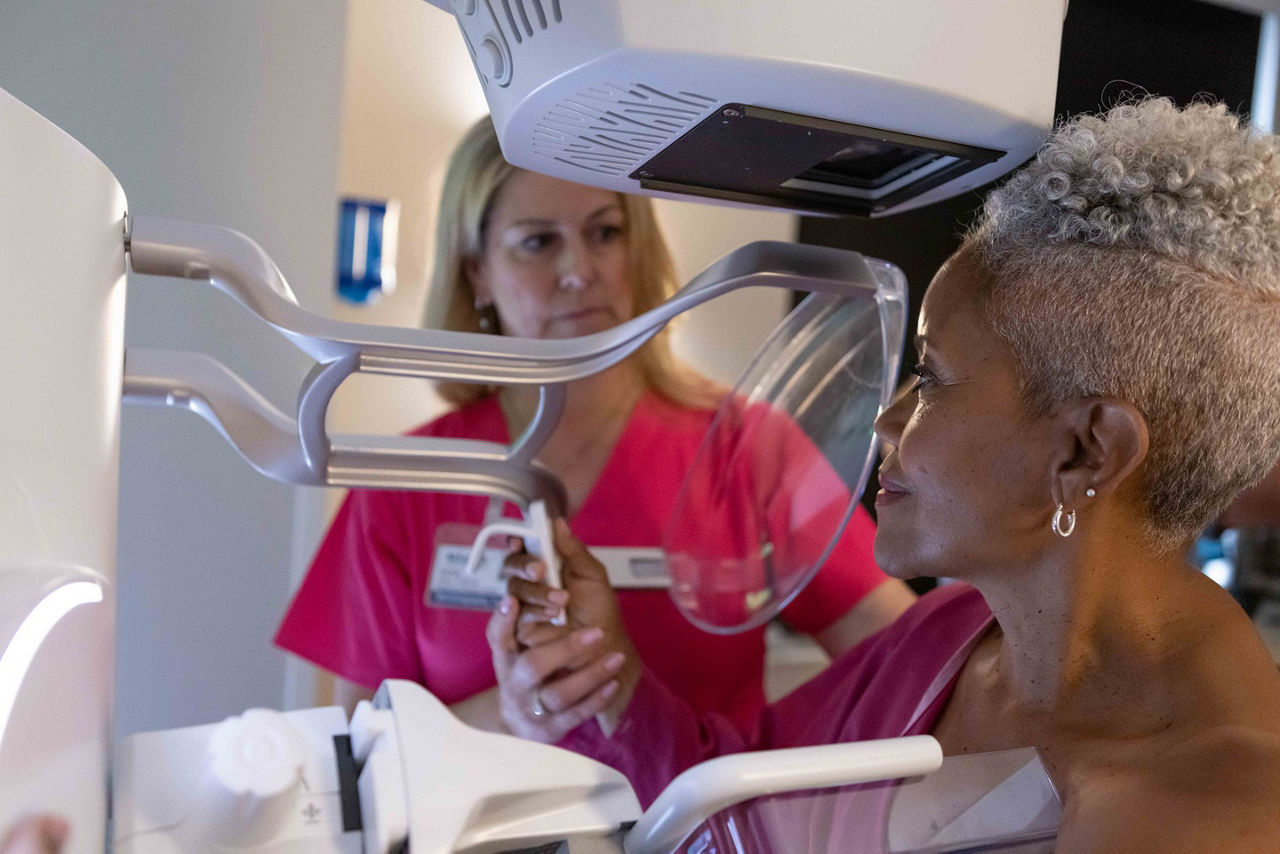
Omnipaque now approved for contrast-enhanced mammography (CEM) as an adjunct to mammography or as an alternative to magnetic resonance imaging (MRI) when MRI is contraindicated or unavailable.1
• Reportedly, 20–30 % of breast cancers are missed on mammograms5
• As an adjunct to mammography, CEM could provide additional information and may facilitate detection or differential diagnosis5
• In a retrospective analysis of 168 consecutive patients with 211 breast lesions, Omnipaque presented with sensitivity 94.5% and specificity 83.3%.6
• Enhancement characteristics of breast lesions on CEM, enhances the performance of digital mammography6